
The AMStent® System
A stent designed for the airway, not adapted for it.
Pulmonary obstruction (PO) is a serious complication of cancers.
Untreated POs can lead to life-threatening respiratory failure.
A common complication of lung cancer and metastatic tumors, pulmonary obstructions block airflow in the central airways, trachea or bronchi. In fact, 25% - 30% of patients with lung cancer or cancers that metastasize to the lung can have POs. Management involves airway stenting to improve airflow and relieve symptoms. If untreated, POs can lead to life-threatening respiratory failure.
Our Technology
The AMStent System

Developed with interventional pulmonologists and thoracic surgeons, the AMStent System is the first and only covered stent leveraging a decellularized human amniotic membrane (DCAM) cover.
The AMStent System combines a minimally invasive catheter with a self-expanding nitinol stent covered in DCAM. The DCAM, primarily composed of extracellular matrix (ECM) common to all human tissue, is intended to minimize the body’s natural foreign body response.



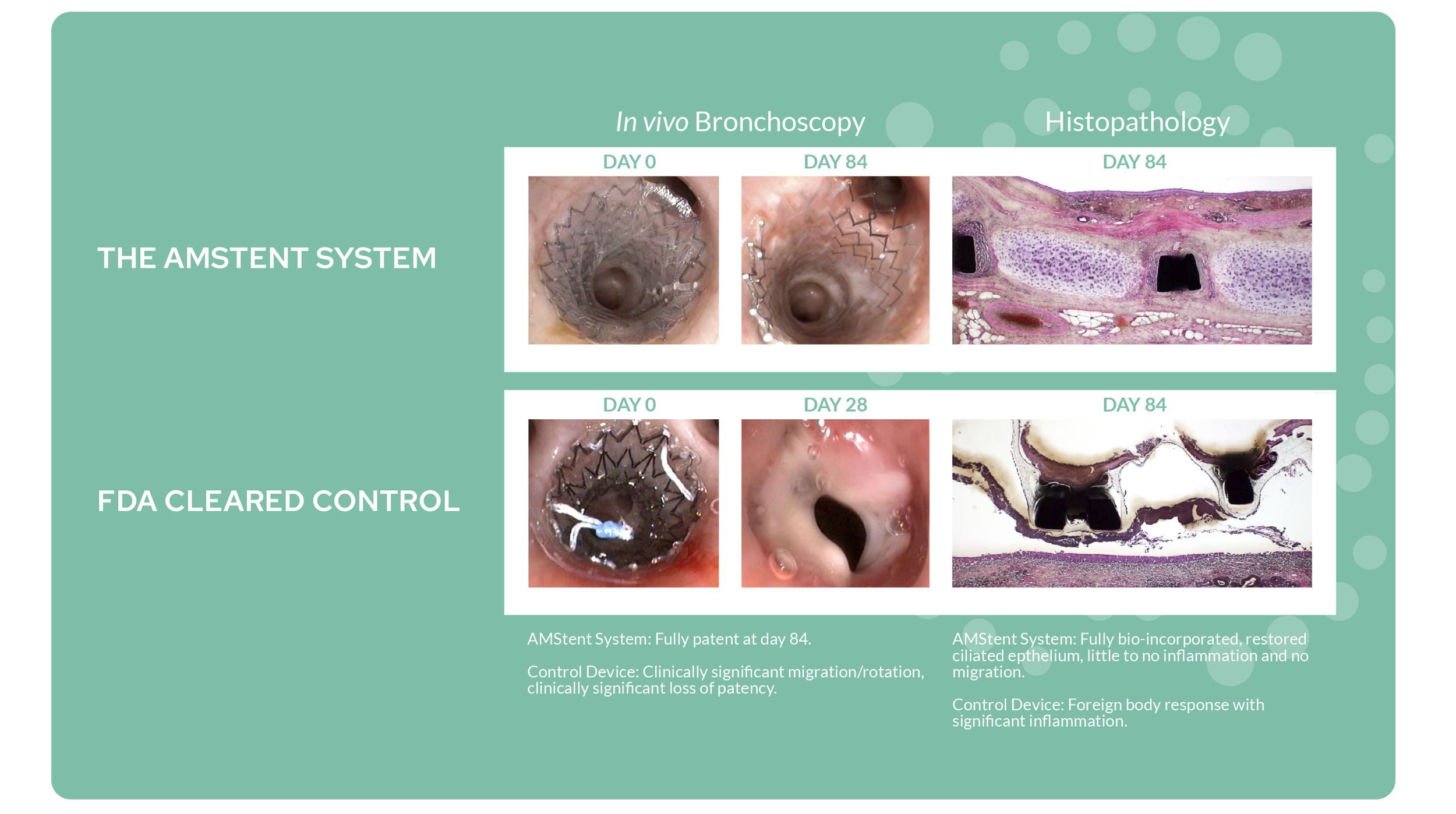
Superior Pre-Clinical Study Results
In chronic animal studies¹, the AMStent System demonstrated improved effectiveness versus a commercially available control stent.
1 On file, Peytant Solutions Chronic GLP Animal Study
